Co-authored by Stanford’s Dr. Carolyn Kaufman and Flosonics Medical CMO Dr. Jon-Emile Kenny, the SaFER framework uses a wearable Doppler patch to help critical care teams determine whether IV fluids will help or harm a sepsis patient in real time, without specialist support
Flosonics Medical, maker of FloPatch, the world’s first wireless, wearable Doppler ultrasound for critical care, today announced the peer-reviewed publication of the Safe and Fluid Effective Resuscitation (SaFER) protocol in SN Comprehensive Clinical Medicine. Co-authored by Flosonics co-founder and CMO Dr. Jon-Emile Kenny and Stanford University’s Dr. Carolyn Kaufman, the paper introduces a structured bedside framework for IV fluid decisions and identifies FloPatch as a practical tool to execute it, including for clinicians without advanced ultrasound training.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260317709165/en/
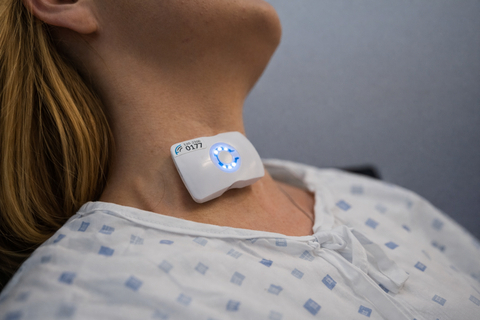
FloPatch, a wearable Doppler ultrasound device, is applied to the neck to measure real-time blood flow and help clinicians make faster, more personalized treatment decisions.
The SaFER protocol structures this decision around three questions: Are fluids indicated? Are fluids safe? Are fluids effective? Existing ultrasound tools can answer these questions but require advanced training most frontline clinicians don’t have. FloPatch bridges that gap. A single wireless patch placed on the neck simultaneously assesses fluid safety and fluid efficacy, providing the same physiologic insight as more complex methods without specialized expertise.
“In rapid response situations, fluid decisions need to be made quickly and with confidence, but the physiologic data that should guide those decisions has historically been out of reach for most frontline teams,” says Dr. Carolyn Kaufman, MD, PhD, Department of Medicine, Stanford University School of Medicine. “The SaFER protocol was designed to change that. The carotid and jugular Doppler data FloPatch provides maps directly onto the questions we’re trying to answer, and any member of the response team can apply it at the bedside.”
“What the SaFER framework makes explicit is that fluid safety and fluid efficacy are physiologically distinct questions,” says Dr. Jon-Emile Kenny, MD, Co-Founder and Chief Medical Officer of Flosonics Medical. “FloPatch was developed specifically for this clinical context, giving physicians and nurses without ultrasound training real-time and easy-to-acquire critical care ultrasound for IV volume management.”
Tailored IV fluid delivery as described by SaFER is important because nearly one-third of patients with septic shock do not respond to additional fluids1, and fluid overload can extend hospital stays by approximately three days at more than $15,000 in added cost per patient2. When clinicians employ dynamic fluid assessments, outcomes improve, with reduced need for mechanical ventilation and renal replacement therapy3,4, shorter ICU stays4, and lower mortality5,6. The Society of Critical Care Medicine recently endorsed ultrasound-guided fluid management for all critically ill patients, but adoption has been limited by the training and equipment demands of traditional tools.
Disclosure: Dr. Jon-Emile Kenny and co-author Joseph Eibl are co-founders of Flosonics Medical, as noted in the published paper’s competing interests statement.
About Flosonics Medical
Flosonics Medical is a medical device company building the future of hemodynamic monitoring. Its flagship product, FloPatch, is an FDA-cleared, wireless, wearable Doppler ultrasound for real-time hemodynamic assessment at the bedside. Recently awarded a Vizient Innovative Technology contract, FloPatch is used in hospitals across the United States to guide fluid resuscitation in critically ill patients. Founded in 2015 in Sudbury, Ontario. For more information, visit www.flosonicsmedical.com.
References:
Sources for statistics and clinical evidence cited in release body:
1. Hernández G, Ospina-Tascón GA, Damiani LP, et al; ANDROMEDA-SHOCK Investigators. Effect of a resuscitation strategy targeting peripheral perfusion status vs serum lactate levels on 28-day mortality among patients with septic shock. JAMA. 2019;321(7):654–664.
2. American Hospital Directory analysis of FFY2022 Medicare IPPS (MedPAR) claims data.
3. Douglas IS, Alapat PM, Corl KA, et al. Fluid response evaluation in sepsis hypotension and shock. Chest. 2020;158(4):1431–45.
4. Latham HE, Bengtson CD, Satterwhite L, et al. Stroke volume guided resuscitation in severe sepsis and septic shock improves outcomes. Journal of Critical Care. 2017;42:42–6.
5. Wang J, Blake LM, Orozco N, et al. Dynamic Measures of Fluid Responsiveness to Guide Resuscitation in Patients With Sepsis and Septic Shock: A Systematic Review and Meta-Analysis. Critical Care Explorations. 2025;7(9):e1303.
6. Sharif S, Flindall H, Basmaji J, et al. Critical Care Ultrasonography for Volume Management: A Systematic Review, Meta-Analysis, and Trial Sequential Analysis of Randomized Trials. Crit Care Explor. 2025;7(5):e1261.
Notes to Editors:
Publication details and supporting references for journalist use:
Full citation: Kaufman CS, Wu D, Eibl JK, Kenny JES. Ultrasonographic Measures of Safe and Fluid Effective Resuscitation (SaFER): Incorporating the VExUS and VTI into Protocolized Care. SN Compr Clin Med. 2026;8:73. DOI: 10.1007/s42399-026-02318-7
SCCM guideline reference: Díaz-Gómez JL, et al. Society of Critical Care Medicine Guidelines for the Use of Point-of-Care Ultrasound in the Adult Critically Ill Patient: 2024 Focused Update. Crit Care Med. 2025;53(2):e447–458.
View source version on businesswire.com: https://www.businesswire.com/news/home/20260317709165/en/
Contacts
Media Contact:
Jamie Gray, Flosonics Medical, press@flosonicsmedical.com





